Chemical Properties of Chlorine - Chemistry of Chlorination
Chemical Properties of Chlorine - Chemistry of Chlorination
Here are some important chemical properties of chlorine:
Reactivity:
Chlorine is a highly reactive element, readily forming compounds with other elements. It has a strong tendency to gain one electron to achieve a stable electron configuration, resulting in the formation of chloride ions (Cl-). Chlorine can react with a wide range of substances, including metals, nonmetals, and organic compounds.
Oxidizing Agent:
Chlorine is a powerful oxidizing agent. It has a high affinity for electrons, making it capable of oxidizing other substances by accepting electrons from them. For example, chlorine can react with metals to form metal chlorides while gaining electrons in the process.
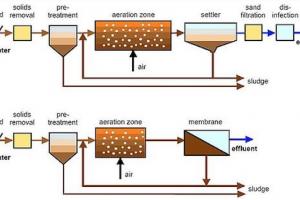
Disinfectant and Bleaching Agent:
Chlorine's strong oxidizing properties make it an effective disinfectant and bleaching agent. It is widely used to sanitize drinking water, swimming pools, and wastewater treatment systems. Chlorine can kill bacteria, viruses, and other microorganisms by disrupting their cellular structures.
Reactivity with Hydrogen:
Chlorine readily reacts with hydrogen to form hydrogen chloride (HCl), a highly corrosive gas. This reaction is exothermic and releases a large amount of heat. Hydrogen chloride is soluble in water, forming hydrochloric acid.
Reaction with Organic Compounds:
Chlorine reacts with various organic compounds, often leading to substitution reactions. It can replace hydrogen atoms in hydrocarbons and other organic molecules. These reactions, known as chlorination reactions, are widely utilized in the production of plastics, pesticides, and other industrial chemicals.
Reaction with Water:
When chlorine gas is dissolved in water, it undergoes a reaction to form a mixture of hypochlorous acid (HOCl) and hydrochloric acid (HCl). This mixture is known as chlorine water and is commonly used as a disinfectant.
Polymerization:
Chlorine can participate in polymerization reactions, leading to the formation of polyvinyl chloride (PVC) and other chlorinated polymers. PVC is a versatile plastic widely used in construction, electrical insulation, and other applications.
Free Available Chlorine
Effective chlorine disinfection depends upon its chemical form in water. The influencing factors are pH, temperature, & organic content in water. When chlorine gas dissolved in water, it rapidly hydrolysis to hydrochloric acid (HCL) & hypochlorous acid (HOCL)
Cl2 + H2O ? H+ + Cl- + HOCL
The equilibrium constant is KH = [H+] [Cl-] [HOCL]
[Cl2 (aq)] = 4.48 × 10 - 4 at 25 °C
Hypochlorous acid is a weak acid & subject to the further dissociation of hypochlorite ions & hydrogen ions
HOCL? OCl- + H+
& its acid dissociation constant Ka is Ka = [OCl-] [H+]
HOCL = 3.7 × 10-8 at 25 ?C
Combine Available Chlorine
Chlorine reacts with certain dissolved constituents in water, such as ammonia & amino nitrogen compounds to produce the chloramines. These are referred to as combined chlorine. In the presence of ammonium ions, free chlorine reacts in a stepwise manner to form three species:
- Monochloramine NH2Cl
- Dichloramine NHCl
- Trichloramine NCl3